 In this example, the nitrogen and each hydrogen has a formal charge of zero. The middle structure: Similarly, we calculate the. The sum of the formal charges of each atom must be equal to the overall charge of the molecule or ion. Molecular Structure The top structure: First, we calculate the formal charge of the nitrogen on the left. It is used to predict the correct placement of electrons. It reflects the electron count associated with the atom compared to the isolated neutral atom. 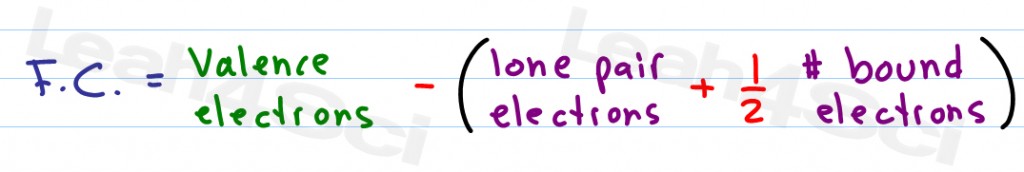 
FC V ( LP +.5 BE) Now that we know the formula let us look at the example of how to find out formal charges for individual atoms in a polyatomic molecule. If nitrite ions do indeed contain a single and a double bond, we would expect for the two bond lengths to be different.\] Formal charge is the charge assigned to an atom in a molecule, assuming that electrons in all chemical bonds are shared equally between atoms. One can calculate the formal charges for any given atom with the help of the following formula: F.C Valence electrons Nonbonding electrons- Bonding electrons/2. 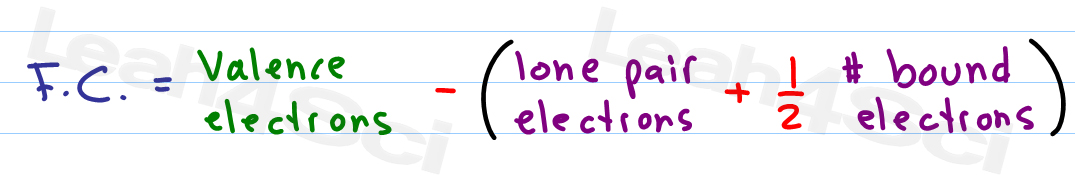
Brackets surround this structure, and there is a superscripted negative sign. So all you do to calculate formal charge is you take the group number, whatever that is, that could be Group four, Group five, whatever. The right structure shows an oxygen atom with two lone pairs of electrons double bonded to a nitrogen atom with one lone pair of electrons that is single bonded to an oxygen atom with three lone pairs of electrons. 1974, the Patent Office in calculating the balance of issue fee due. Brackets surround this structure, and there is a superscripted negative sign. PATENT OFFICE NOTICES Availability of Rules of Practice A new edition of ' 37 Code. In order to use the formula charge formula, we must observe the Lewis Dot Structure for. There is neither deficiency nor excess of electrons with Be in the lewis structure. The left structure shows an oxygen atom with three lone pairs of electrons single bonded to a nitrogen atom with one lone pair of electrons that is double bonded to an oxygen with two lone pairs of electrons. Step 1: Draw the Lewis Dot Structure for the compound given in order to calculate the formal charge. Be has 2 valence electrons which are shared by covalent bonding. The electrons involved in the N–O double bond, however, are in different positions: 
\) can have two possible structures with the atoms in the same positions. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
